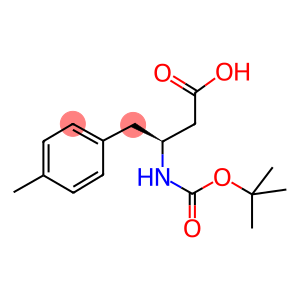
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid
CAS: 270062-96-9
Molecular Formula: C16H23NO4
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid - Names and Identifiers
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid - Physico-chemical Properties
| Molecular Formula | C16H23NO4 |
| Molar Mass | 293.36 |
| Storage Condition | 2-8°C |
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 10 |
| Hazard Class | IRRITANT |
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid - Introduction
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric is an organic compound whose chemical formula is C16H23NO4. The following is a description of some of its properties, uses, methods and safety information:
Nature:
1. Appearance: usually white crystalline solid;
2. melting point: about 120-125 degrees Celsius;
3. Stability: Stable at room temperature, soluble in some organic solvents, such as dimethyl sulfoxide (DMSO), dichloromethane, etc;
4. optical activity: Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid has chirality, where (S) indicates that it is a levorotatory isomer.
Use:
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid is commonly used in the field of organic synthesis as an intermediate for the preparation of chiral compounds. It is a common amino acid protecting group, by adding it to the molecule, the amino acid can be kept stable during the synthesis process.
Preparation Method:
The preparation of Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid usually includes the following steps:
1. First, 3-amino -4-(4-methylphenyl) butyric acid was synthesized by chemical reaction;
2. Then, it reacts with Boc(tert-butoxycarbonyl) group to generate Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid.
Safety Information:
1. Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid is not a hazardous chemical, but it still needs to comply with the basic laboratory safety operation specifications;
2. In the process of use, avoid contact with strong oxidants, strong acids and other harmful substances to avoid dangerous reactions;
3. May be irritating to the eyes, skin and respiratory tract, so wear appropriate personal protective equipment when using.
Nature:
1. Appearance: usually white crystalline solid;
2. melting point: about 120-125 degrees Celsius;
3. Stability: Stable at room temperature, soluble in some organic solvents, such as dimethyl sulfoxide (DMSO), dichloromethane, etc;
4. optical activity: Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid has chirality, where (S) indicates that it is a levorotatory isomer.
Use:
Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid is commonly used in the field of organic synthesis as an intermediate for the preparation of chiral compounds. It is a common amino acid protecting group, by adding it to the molecule, the amino acid can be kept stable during the synthesis process.
Preparation Method:
The preparation of Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid usually includes the following steps:
1. First, 3-amino -4-(4-methylphenyl) butyric acid was synthesized by chemical reaction;
2. Then, it reacts with Boc(tert-butoxycarbonyl) group to generate Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid.
Safety Information:
1. Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid is not a hazardous chemical, but it still needs to comply with the basic laboratory safety operation specifications;
2. In the process of use, avoid contact with strong oxidants, strong acids and other harmful substances to avoid dangerous reactions;
3. May be irritating to the eyes, skin and respiratory tract, so wear appropriate personal protective equipment when using.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (S)-4-Methyl-b-(Boc-amino)benzenebutanoic acid Visit Supplier Webpage Request for quotationCAS: 270062-96-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid Request for quotation
CAS: 270062-96-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 270062-96-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BOC-(S)-3-AMINO-4-(4-METHYL-PHENYL)-BUTYRIC ACID Request for quotation
CAS: 270062-96-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 270062-96-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Boc-(S)-3-Amino-4-(4-Methylphenyl)Butanoic Acid Visit Supplier Webpage Request for quotation
CAS: 270062-96-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 270062-96-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-4-Methyl-b-(Boc-amino)benzenebutanoic acid Visit Supplier Webpage Request for quotationCAS: 270062-96-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boc-(S)-3-Amino-4-(4-methyl-phenyl)-butyric acid Request for quotation
CAS: 270062-96-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 270062-96-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BOC-(S)-3-AMINO-4-(4-METHYL-PHENYL)-BUTYRIC ACID Request for quotation
CAS: 270062-96-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 270062-96-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Boc-(S)-3-Amino-4-(4-Methylphenyl)Butanoic Acid Visit Supplier Webpage Request for quotation
CAS: 270062-96-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 270062-96-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



